THE KARIUS SPECTRUM TEST CAN HELP YOU COMPLETE THE PNEUMONIA PUZZLE
Diagnostic Application: Pneumonia
Bronchoscopy can fail to yield a diagnosis in 50% - 75% of procedures in immunocompromised patients.1-4
Featured Case
Study Site: OSF Saint Francis Medical Center5
39 years old with untreated HIV infection and respiratory failure requiring mechanical ventilation with an unclear etiology.
Tests Performed: Diagnostic tests performed for evaluation of pneumonia and other etiologies included absolute CD4 count of 6, HIV viral load of 332,909 copies/ml, CMV IgG positive; CXR with bilateral multinodular changes, chest CT with miliary lesions, brain MRI with multiple ring enhancing lesions; negative testing included: blood and BAL cultures; VZV, syphilis, and tuberculosis studies; Histoplasma/ Aspergillus/ Legionella/ Cryptococcus/ Influenza antigens.
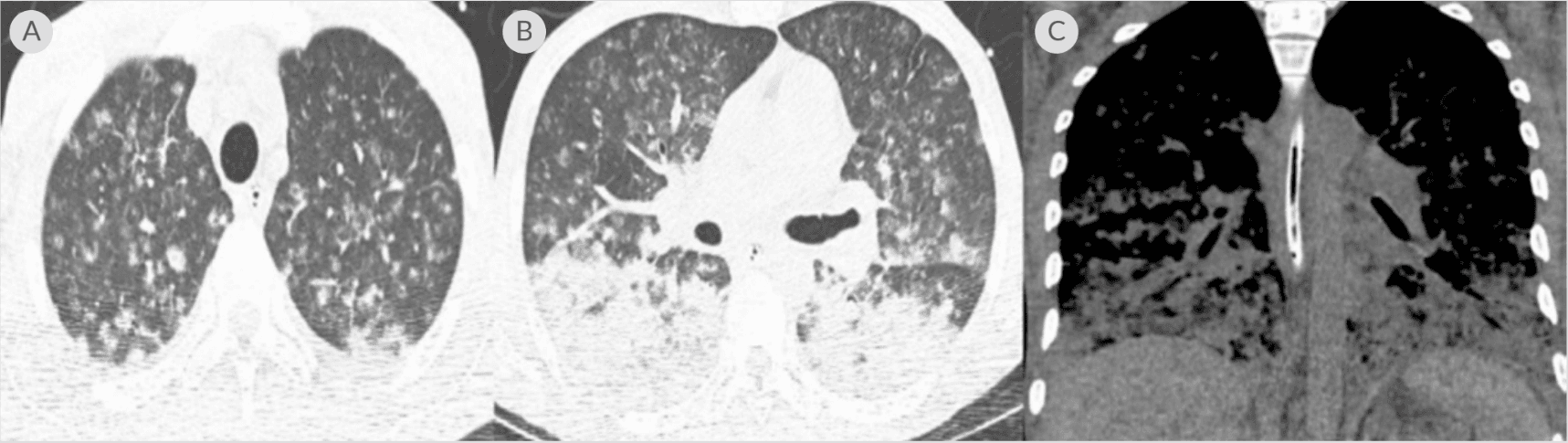
(A) CT chest axial lung window showing miliary changes in both lungs; (B) CT chest axial lung window showing several nodules with developing central cavitation and bilateral consolidation; (C) coronal window showing bibasilar airspace disease with confluent consolidation throughout the lung bases5
Karius Spectrum Test Result: Toxoplasma gondii
Outcome: Started 6-week course of pyrimethamine/sulfadiazine, discontinued piperacillin/tazobactam; resulting in significant clinical improvement allowing the patient to be started on antiretroviral therapy.
Clinical Evidence
Study Site: 10 Medical Centers*
In a prospective, multi-center study enrolling over 173 adult patients with suspected pneumonia, a 40% (21/52) relative increase in the cause of pneumonia identified when the Karius Spectrum test was used in conjunction with standard of care (SOC) testing.**,6
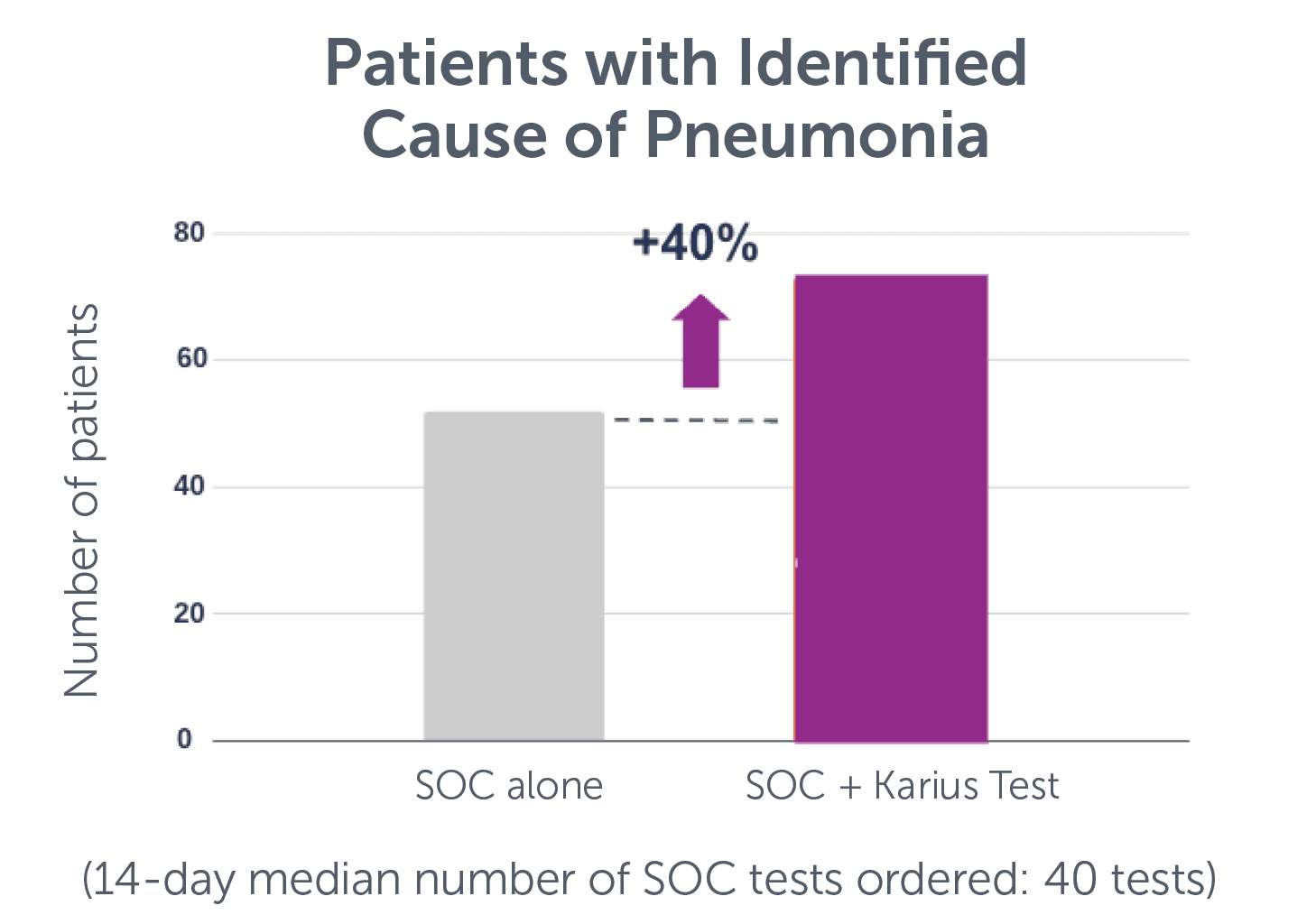
Moreover, the primary endpoint was achieved: 1 in 6 patients (21/121) had a cause of pneumonia identified via the Karius Spectrum test where a thorough SOC of 15+ tests did not. 81% (17/21) of these patients would have had an adjudicated possible antimicrobial change with the Karius Spectrum test result where SOC failed.**,6
*Please see PICKUP Study Summary for full list of enrolling centers
**Please see study for comparable tests
_____________________________________________
Study Site: Rady Children’s Hospital
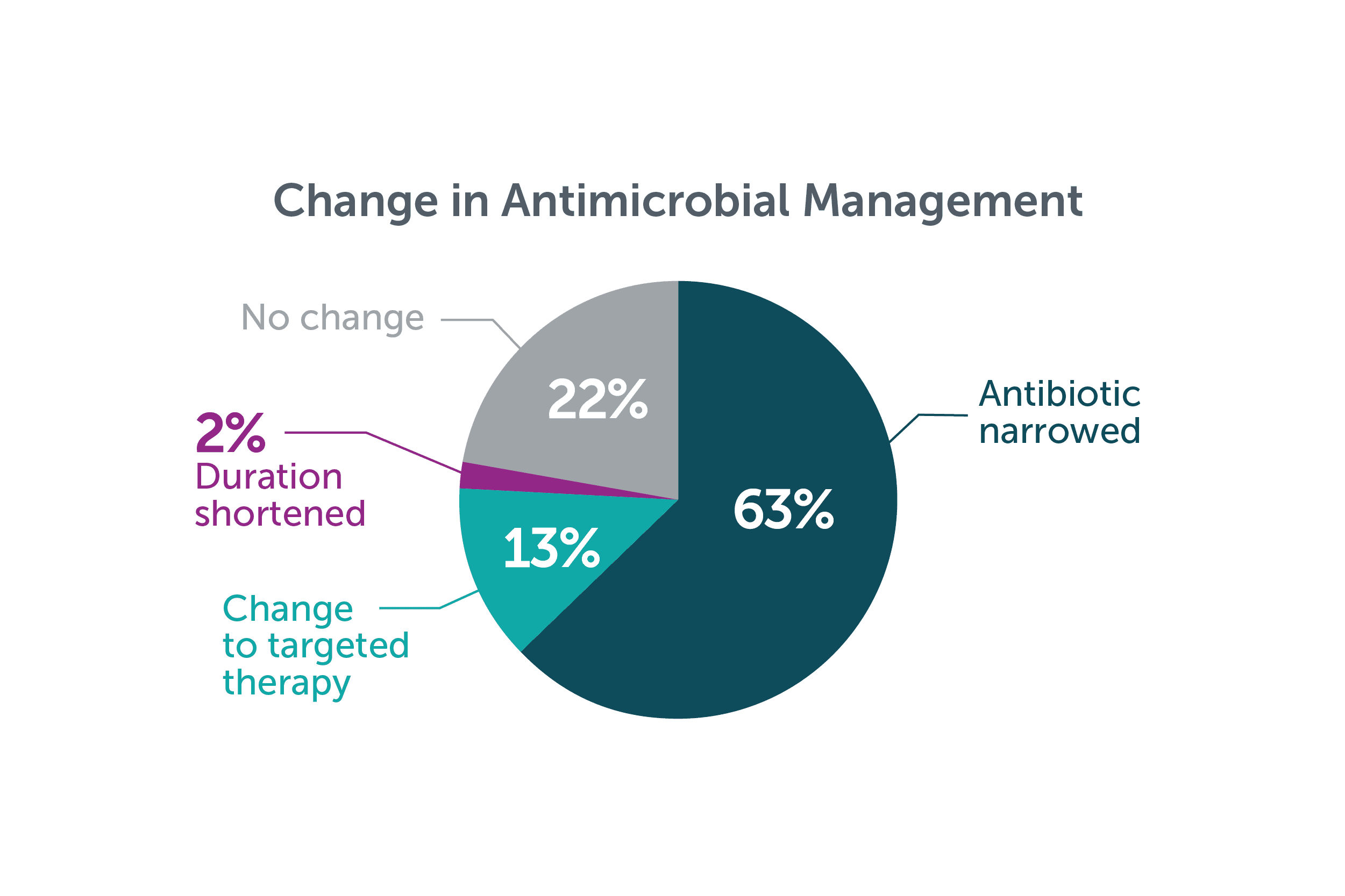
In a retrospective study in 46 immunocompetent pediatric patients with complicated community acquired pneumonia, the Karius Spectrum test led to a change in antimicrobial management in 78% (35/46).7
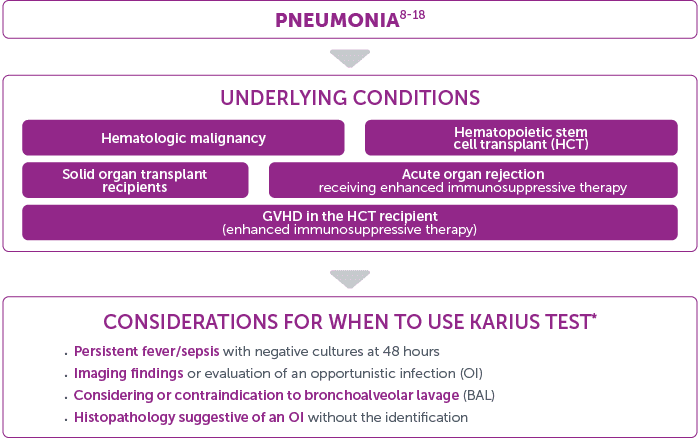
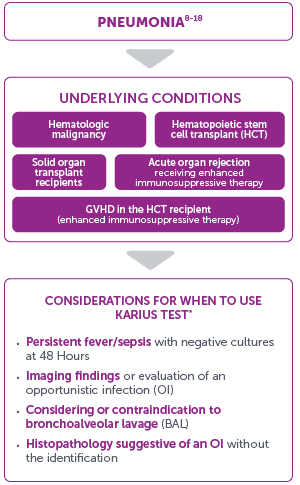
Considerations for Use

Learn More About the Karius Spectrum
*Karius Spectrum is an LDT. Clinicians should independently evaluate its use and interpret test results. This content is provided for informational purposes only and is not intended or implied to be an exhaustive list of consideration or substitute for existing medical expertise or guideline recommendations.
References:
1. Bauer PR et al. Eur Respir J. 2019;54(1).
2. Batra S et al. Pediatr Blood Cancer. 2015;62(9):1579-1586.
3. Hofmeister CC et al. Bone Marrow Transplant. 2006;38(10):693-698
4. Patel NR et al. Chest. 2005;127(4):1388-1396.
5. Roy M, et al. Eur J Case Rep Intern Med. 2020 Sep 4;7(11):001862.
6. Bergin, SP et al, Plasma Microbial Cell-Free DNA Sequencing in Immunocompromised Patients with Pneumonia: A Prospective Observational Study, Clinical Infectious Diseases, 2023;, ciad599, https://doi.org/10.1093/cid/ciad599
7. Dworsky ZD, et al. Hosp Pediatr. 2022 Apr 1;12(4):377-384.
8. Shishido AA, et al. BMC Infect Dis. 2022 Apr 13;22(1):372.
9. Husain S, et al. Clin Transplant. 2019 Sep;33(9):e13544.
10. Restrepo A, et al. Clin Transplant. 2019 Sep;33(9):e13509.
11. Fishman JA, et al. Clin Transplant. 2019 Sep;33(9):e13587.
12. Dulek DE, et al. Clin Transplant. 2019 Sep;33(9):e13545.
13. Hill JA, et al. Clin Infect Dis. 2021 Dec 6;73(11):e3876-e3883.
14. Azar MM, et al. Am J Transplant. 2022 Dec;22(12):3150-3169.
15. NCCN. Prevention and treatment of cancer-related infections. Version 2.2022.
16. Ramirez JA, et al. Chest. 2020 Nov;158(5):1896-1911.
17. Certan M, et al. Clin Infect Dis. 2022 Sep 29;75(6):1046-1053.
18. Schuster MG et al. Open Forum Infect Dis. 2017;4(2):ofx050.