

Pneumonia in the ImmunoCompromised: Use of the Karius Test for the Detection of Undiagnosed Pathogens
Introduction
In patients with hematologic malignancies (HM) or those who have undergone hematopoietic cell transplantation (HCT), pneumonia is a common infection and often contributes to mortality.1, 2, 3
Study Design
The PICKUP study is the first multicenter, prospective, observational study designed to compare Karius Test® to Standard Of Care (SOC) in adults with HM or HCT with suspicion of pneumonia.
The PICKUP Study enrolled 257 adult patients across 10 leading U.S. medical centers:
- Duke University Health
- University of Pittsburgh Medical Center
- Fred Hutchinson Cancer Research Center
- MD Anderson Cancer Center
- City of Hope
- Tulane Medical Center
- University of California, San Francisco Medical Center
- University of Colorado Denver
- Weill Cornell Medicine
- Memorial Sloan Kettering Cancer Center
Primary Endpoint:
The primary endpoint of this study was to determine the additive diagnostic value of the Karius Test, defined as the proportion of patients where the SOC was negative that had a probable cause of pneumonia exclusively identified by plasma mcfDNA sequencing
A rigorous Minimum Diagnostic Standard* consisting of both non-invasive and invasive standard of care (SOC) testing was required for study inclusion:
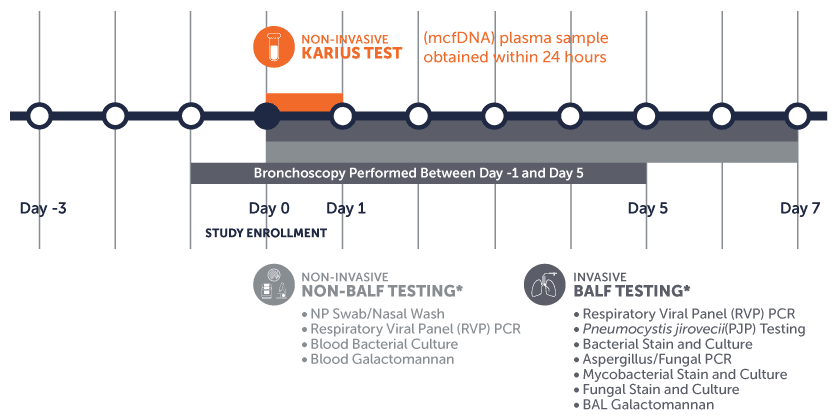


* Reported within 7 days of enrollment
** Note all other microbiological testing (BALF and non-BALF) typically conducted at the respective institution was included, in addition to the Minimum Diagnostic Standard
Results4
Pathogens causing pneumonia were often not detected by SOC.
30% identification of pneumonia causing pathogens by invasive or non-invasive SOC in the PICKUP Study (15+ tests on average)
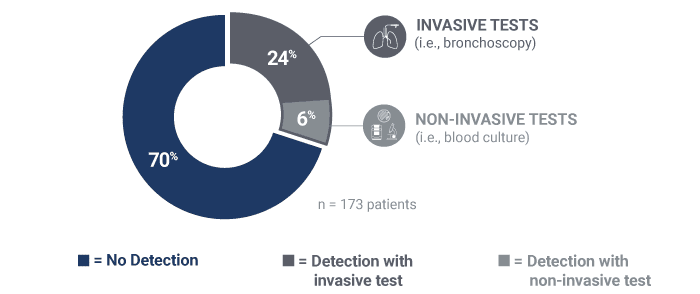
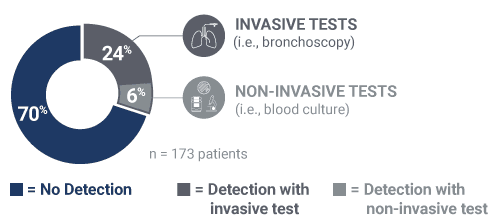
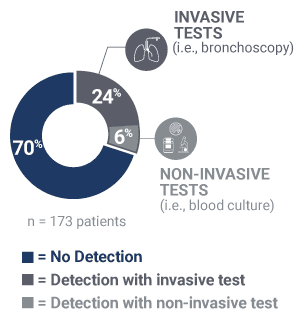
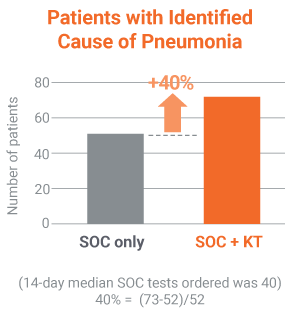
40% relative increase in the adjudicated cause of pneumonia identified when the Karius Test was added to SOC

Pathogens causing pneumonia identified exclusively by Karius Test
Possible antimicrobial change in 81% (17/21) of patients with a pneumonia etiology identified exclusively by Karius Test8
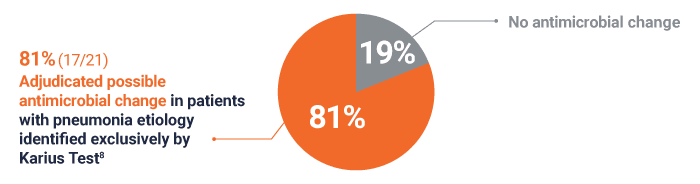
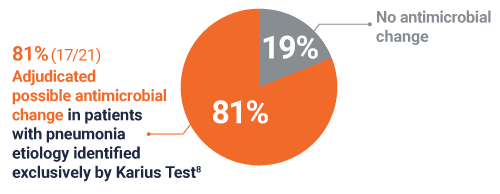

For 17 patients, adjudicated possible antimicrobial changes included: broadening of, narrowing of, and/or earlier antibacterial therapy (12/17), antiviral therapy changes (1/17), and antifungal coverage changes (5/17)
Pneumonia pathogens uniquely identified by the Karius Test
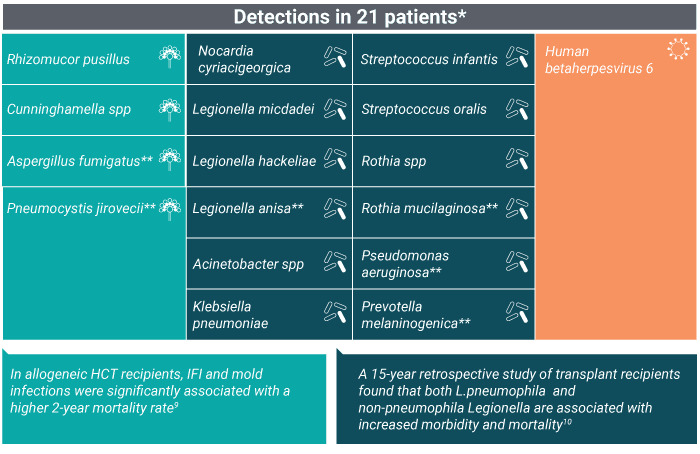
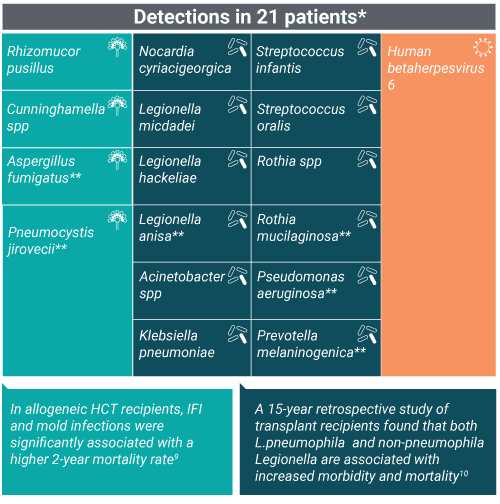
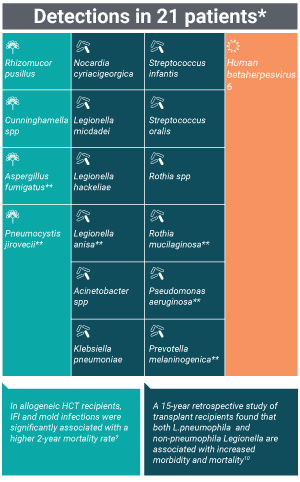
*Three patients had two adjudicated probable causes of pneumonia detected by plasma mcfDNA sequencing
**Note that 6 of these microbes were identified as pneumonia etiology in multiple patients
Conclusion
Adding the Karius Test to the diagnostic workup for identifying causative pathogens in pneumonia can improve diagnostic yield over SOC alone in patients with hematological malignancies or HCT.
If you would like more information about this study, please fill form below:
References:
- Certan M, et al. Clin Infect Dis. 2022;75(6):1046–1053
- Schuster MG, et al. Open Forum Infect Dis. 2017;4(2):ofx050
- Batra S et al. Pediatr Blood Cancer. 2015;62(9):1579-1586
- Bergin, SP et al, Plasma Microbial Cell-Free DNA Sequencing in Immunocompromised Patients with Pneumonia: A Prospective Observational Study, Clinical Infectious Diseases, 2023;, ciad599, https://doi.org/10.1093/cid/ciad599