THE KARIUS SPECTRUM TEST CAN HELP YOU COMPLETE THE ENDOCARDITIS PUZZLE
Diagnostic Application: Endocarditis
Blood culture-negative endocarditis can be as high as 10% to 40%.1,2
Featured Case
Study Site: Weill Cornell Medicine
29 year old male with a history of congenital heart disease requiring multiple cardiac surgeries, including exposure to a Heater-Cooler device that was associated with a Mycobacterium chimaera outbreak.
Echocardiogram revealed a pulmonary valve echodensity suggestive of a vegetation.
Tests Performed: Blood cultures (negative); Serologies for B. henselae, Brucella species, C. burnetii, and other atypical pathogens were performed, with a positive result for C. burnetii after one week.
Karius Spectrum Test Result: C. burnetii, within 48 hours, before serology results
Outcome: Earlier diagnosis leading to initiation of targeted therapy, with complete resolution of symptoms after 4 weeks of therapy.
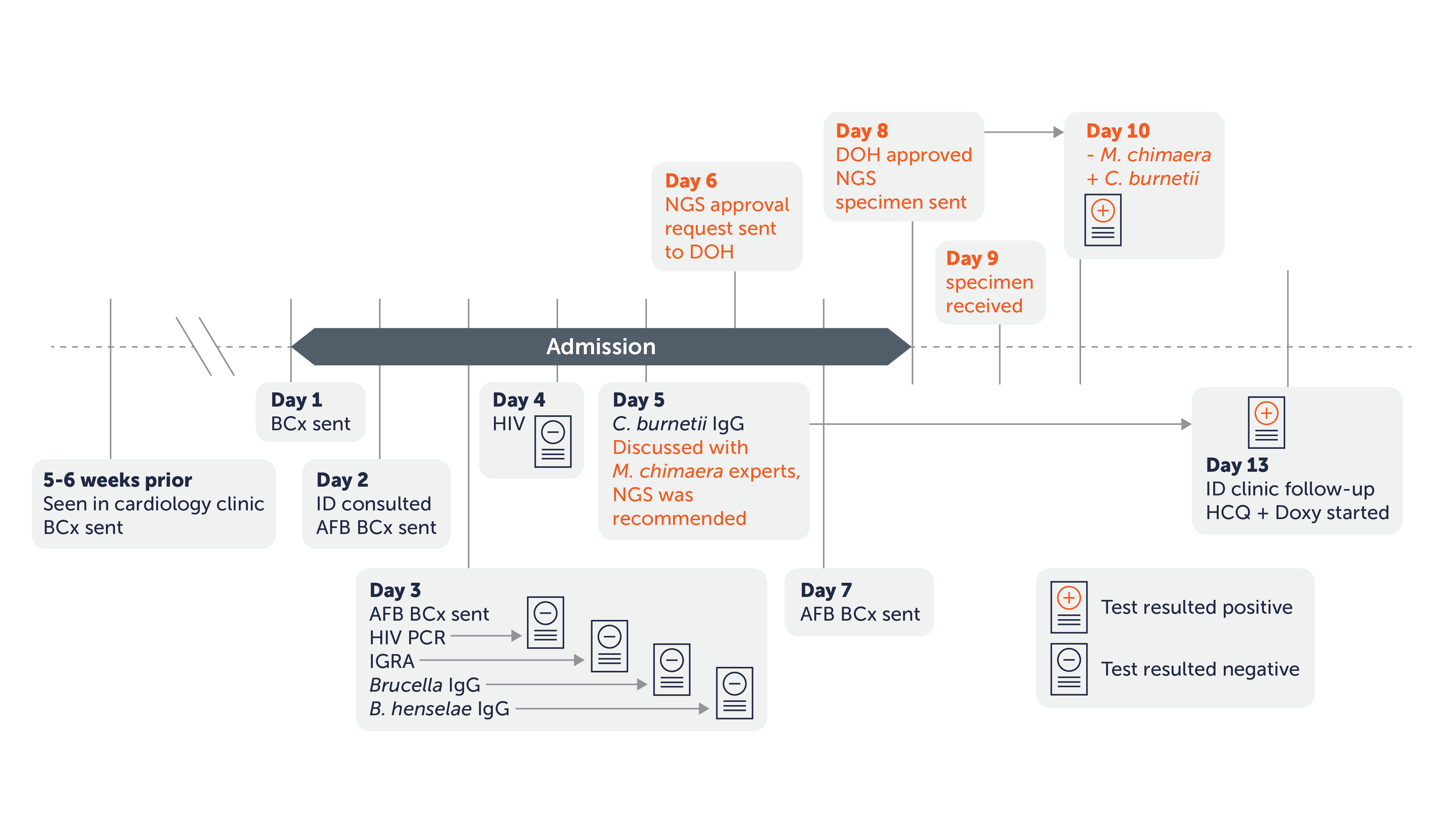
A. Timeline of microbiologic testing. All microbiologic assays (with the exception of plasma mcfDNA NGS) were recommended during the initial infectious diseases consultation (day 2). The day when each assay was sent is shown in the timeline. The position of the result icon correlates with the day each result became available. The timeline of plasma mcfDNA NGS testing is highlighted in orange-colored text.
Karius Spectrum Test in 2023 Duke-ISCVID Criteria for Infective Endocarditis
First time inclusion of hypothesis-free metagenomic testing, such as the Karius Spectrum test, for the rapid identification of hard-to-detect pathogens, specifically:
- Detection of C. burnetii, Bartonella species, or T. whipplei (Major Criteria)
- Detection of other microbes consistent consistent with IE (Minor Criteria)10
Clinical Evidence
Study Site: Duke University
In 23 adult patients with definite endocarditis, the median duration of positivity from antibiotic treatment initiation was estimated to be approximately 38.1 days for mcfDNA versus 3.7 days for blood culture.4

_____________________________________________
Study Site: Rady Children’s Hospital
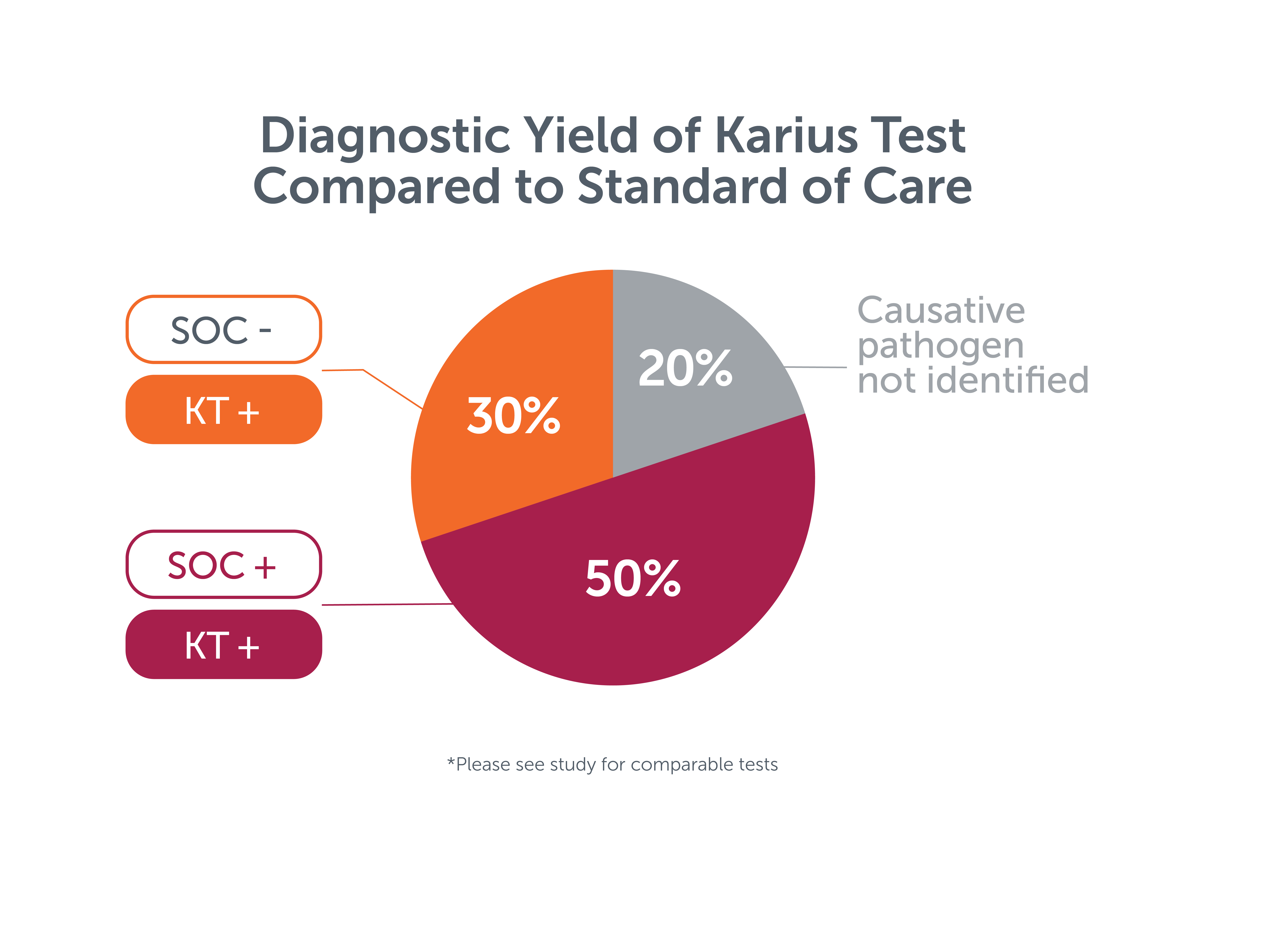
In a case series of 10 pediatric patients with endocarditis, blood cultures were positive in 3 patients and valve broad range PCR was positive in 2 patients for a combined yield of 50%. The Karius Spectrum test identified the causative pathogen in 8 patients (80%), including 3 cases in which standard testing* failed.5
Considerations for Use
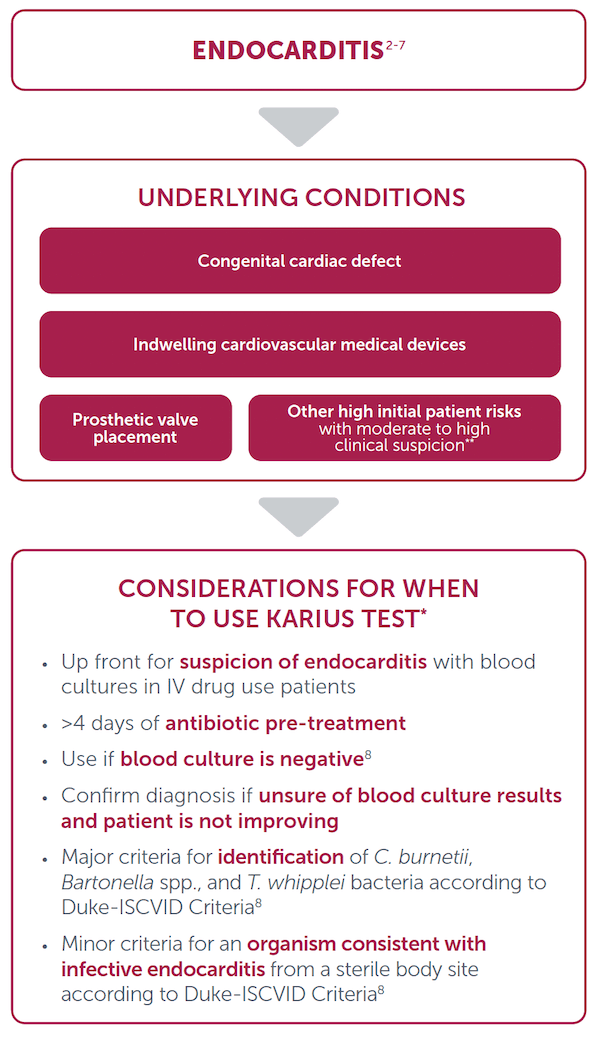
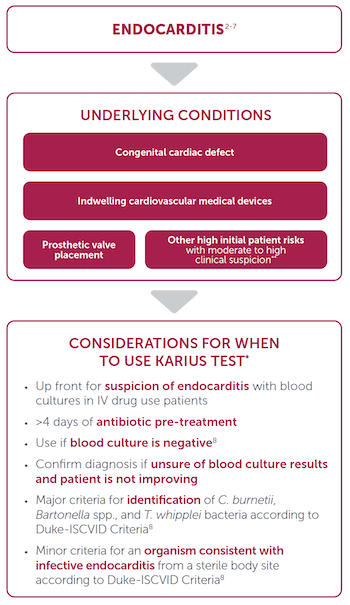

*Karius Spectrum is an LDT. Clinicians should independently evaluate its use and interpret test results. This content is provided for informational purposes only and is not intended or implied to be an exhaustive list of consideration or substitute for existing medical expertise or guideline recommendations.
**Other high initial patient risks include previous endocarditis, new murmur, heart failure, or other stigmata of endocarditis.9
Learn More About the Karius Spectrum
References:
1. Murdoch DR, et al. Arch Intern Med. 2009 Mar 9;169(5):463-73.
2. Lamas CC, et al. Infection. 2016 Aug;44(4):459-66. Epub 2015 Dec 15.
3. Kondo M, et al. Open Forum Infect Dis. 2019 Jun 1;6(6):ofz242. eCollection 2019 Jun.
4. Eichenberger EM, et al. Clin Infect Dis. 2022 Jun 10;ciac426. Online ahead of print.
5. To RK, et al. Pediatr Infect Dis J. 2021 May 1;40(5):486- 488.
6. Shishido AA, et al. BMC Infect Dis. 2022 Apr 13;22(1):372.
7. Solanky D, et al. Front. Trop. Dis. 3:842100.
8. Fowler VG, et al. Clin Infect Dis. 2023 May 4;ciad271. Online ahead of print.
9. Baddour LM, et al. Circulation. 2015 Oct 13;132(15):1435-86. Epub 2015 Sep 15
10. Fowler VG, et al. Clin Infect Dis. 2023 May 4;ciad271. Online ahead of print.